|
Nucleic acid and its related compounds, such as pyrimidines and purines, are well known to absorb UV light at a wavelength of 260 nm
Ohtsuka K1, Osakabe MM.Author informationAbstractThe herbivorous spider mite Tetranychus urticae usually remains on the lower leaf surfaces of its host plants. Although terrestrial animals are generally thought to be well protected from damage because of UV radiation, insect herbivory frequently increases when solar UV-B (280-315 nm) radiation is attenuated. As UV transmission through leaves is generally low because of the accumulation of compounds that act as selective sunscreens (e.g., phenolics), we hypothesized that T. urticae avoids solar UV-B radiation by staying on lower leaf surfaces. We examined whether artificial UV irradiation and solar UV affected the survival and reproduction of T. urticae and whether staying on lower leaf surfaces was beneficial to their performance under ambient UV radiation. We found that T. urticae was not well protected from UV-B radiation, because artificial UV-B irradiation strongly decreased survivorship and egg production. More importantly; compulsory solar UV irradiation treatments also had lethal effects on T. urticae, whereas the mites could avoid them if they remained on the lower leaf surfaces of their host plants. These results showed that access to habitats protected from sunlight, such as lower leaf surfaces, is likely essential for T. urticae survival under ambient UV-B radiation. The lethal effects of solar UV radiation may also affect the population dynamics of spider mites, and habitat (resource) limitation may increase the probability of interspecific interactions, such as competition and predation. In turn, the occurrence of these interactions in sheltered areas may be associated with observed increases in herbivory under conditions of solar UV-B-attenuation. Screening, in medicine, is a strategy used in a population to identify the possible presence of an as-yet-undiagnoseddisease in individuals without signs or symptoms. This can include individuals with pre-symptomatic or unrecognized symptomatic disease. As such, screening tests are somewhat unique in that they are performed on persons apparently in good health.
Screening interventions are designed to identify disease in a community early, thus enabling earlier intervention and management in the hope to reduce mortality and suffering from a disease. Although screening may lead to an earlier diagnosis, not all screening tests have been shown to benefit the person being screened; overdiagnosis, misdiagnosis, and creating a false sense of security are some potential adverse effects of screening. For these reasons, a test used in a screening program, especially for a disease with low incidence, must have good sensitivity in addition to acceptablespecificity.[1] Several types of screening exist: universal screening involves screening of all individuals in a certain category (for example, all children of a certain age). Case finding involves screening a smaller group of people based on the presence of risk factors (for example, because a family member has been diagnosed with a hereditary disease). Screening interventions are not designed to be diagnostic, and often have significant rates of both false positive and false negative results. Contents [hide]
Principles of screening[edit]In 1968 the World Health Organization published guidelines on the Principles and practice of screening for disease, which often referred to as Wilson's criteria.[2]The principles are still broadly applicable today:
Synthesis of emerging screening criteria proposed over the past 40 years • The screening programme should respond to a recognized need. • The objectives of screening should be defined at the outset. • There should be a defined target population. • There should be scientific evidence of screening programme effectiveness. • The programme should integrate education, testing, clinical services and programme management. • There should be quality assurance, with mechanisms to minimize potential risks of screening. • The programme should ensure informed choice, confidentiality and respect for autonomy. • The programme should promote equity and access to screening for the entire target population. • Programme evaluation should be planned from the outset. • The overall benefits of screening should outweigh the harm. [3] Types of screening[edit] A mobile clinic used to screen coal miners at risk of black lung disease
Medical equipment used in screening[edit]Medical equipment used in screening tests is usually different from equipment used in diagnostic tests as screening tests are used to indicate the likely presence or absence of a disease or condition in people not presenting symptoms; while diagnostic medical equipment is used to make quantitative physiological measurements to confirm and determine the progress of a suspected disease or condition. Medical screening equipment must be capable of fast processing of many cases, but may not need to be as precise as diagnostic equipment. Limitations of screening[edit]Screening can detect medical conditions at an early stage before symptoms present while treatment is more effective than for later detection. In the best of cases lives are saved. Like any medical test, the tests used in screening are not perfect. The test result may incorrectly show positive for those without disease (false positive), or negative for people who have the condition (false negative). Limitations of screening programmes can include:
Analysis of screening[edit]To many people, screening instinctively seems like an appropriate thing to do, because catching something earlier seems better. However, no screening test is perfect. There will always be the problems with incorrect results and other issues listed above. Before a screening program is implemented, it should ideally be looked at to ensure that putting it in place would do more good than harm. The best studies for assessing whether a screening test will increase a population's health are rigorous randomized controlled trials. When studying a screening program using case-control or, more usually, cohort studies, various factors can cause the screening test to appear more successful than it really is. A number of different biases, inherent in the study method, will skew results. Screening can certainly improve outcomes, but this must be confirmed with proper statistical analysis, not simplistic comparison of numbers. Lead time bias[edit]For more details on this topic, see Lead time bias. Lead time bias leads to longer perceived survival with screening, even if the course of the disease is not alteredThe intention of screening is to diagnose a disease earlier than it would be without screening. Without screening the disease may be discovered later, when symptoms appear. Even if in both cases a person will die at the same time, because we diagnosed the disease earlier with screening thesurvival time since diagnosis is longer with screening; but life span has not been prolonged, and there will be added anxiety as the patient must live with knowledge of the disease for longer. Looking at statistics of survival time since diagnosis, screening will show an increase (this gain is called lead time). If we do not think about what survival time actually means in this context, we might attribute success to a screening test that does nothing but advance diagnosis; comparing statistics of mortality due to a disease in a screened and unscreened population gives more meaningful information. Length time bias[edit]For more details on this topic, see Length time bias. Length time bias leads to better perceived survival with screening, even if the course of the disease is not altered.Many screening tests involve the detection of cancers. It is often hypothesized that slower-growing tumors have better prognoses than tumors with high growth rates. Screening is more likely to detect slower-growing tumors (due to longer pre-clinical sojourn time), which may be less deadly. Thus screening may tend to detect cancers that would not have killed the patient or even been detected prior to death from other causes. Selection bias[edit]For more details on this topic, see Selection bias.Not everyone will partake in a screening program. There are factors that differ between those willing to get tested and those who are not. If people with a higher risk of a disease are more likely to be screened, for instance women with a family history of breast cancer are more likely than other women to join a mammography program, then a screening test will look worse than it really is: negative outcomes among the screened population will be higher than for a random sample. Selection bias may also make a test look better than it really is. If a test is more available to young and healthy people (for instance if people have to travel a long distance to get checked) then fewer people in the screening population will have negative outcomes than for a random sample, and the test will seem to make a positive difference. Overdiagnosis[edit]For more details on this topic, see Overdiagnosis.Screening may identify abnormalities that would never cause a problem in a person's lifetime. An example of this is prostate cancer screening; it has been said that "more men die with prostate cancer than of it".[6] Autopsy studies have shown that a high proportion of elderly men who have died of other causes are found to have had prostate cancer. Aside from issues with unnecessary treatment (prostate cancer treatment is by no means without risk), overdiagnosis makes a study look good at picking up abnormalities, even though they are sometimes harmless. Overdiagnosis occurs when all of these people with harmless abnormalities are counted as "lives saved" by the screening, rather than as "healthy people needlessly harmed by overdiagnosis". Avoidance of bias[edit]The best way to minimise these biases is to use a randomized controlled trial. These need to be very large[clarification needed], and very strict in terms of research procedure. Such studies take a long time and are expensive, but provide the best information for evidence-based medicine. AbstractThe two-spotted spider mite, Tetranychus urticae was exposed to UV-C (250 nm), UV-B (300 nm), and UV-A (350 nm). In non-diapausing females, the median effective doses for 50% mortality plus escape incidence (ED50) were 21 (UV-C) and 104 kJ m−2 (UV-B); those for 50% oviposition rate in continuous darkness-treated mites were 6.2 (UV-C) and 41 kJ m−2 (UV-B). No significant effects of UV-A on mortality and oviposition rate were observed. The ED50 values for UV-B were similar to the natural UV-B observed for 2–5 days in summer when T. urticae inhabits the undersides of leaves. Therefore, T. urticaepossibly uses leaves as a filter to avoid the deleterious effects of UV-B. In diapausing females, low mortality was observed even at high doses of UV radiation, but more than half escaped even at low doses. The orange body color of diapausing females results from accumulation of carotenoids, a scavenger for UV-induced reactive oxygen species; this may explain the low mortality of diapausing females. Diapausing females may overcome the deleterious effects of UV-B during winter in the absence of leaves by emigrating to UV-free environments and by accumulating carotenoids.
Keywords
From Wikipedia, the free encyclopedia Look up diapause in Wiktionary, the free dictionary. Part of a series on Animal dormancy
Diapause, when referencing animal dormancy, is the delay in development in response to regularly and recurring periods of adverse environmental conditions.[1][2] It is considered to be a physiological state of dormancy with very specific initiating and inhibiting conditions. Diapause is a mechanism used as a means to survive predictable, unfavorable environmental conditions, such as temperature extremes, drought, or reduced food availability. Diapause is most often observed in arthropods, especially insects, and in the embryos of many of the oviparousspecies of fish in the order Cyprinodontiformes.[3] (Diapause does not occur in embryos of the viviparous andovoviviparous species of Cyprinodontiformes.) Diapause is not only induced in an organism by specific stimuli or conditions, but once it is initiated, only certain other stimuli are capable of bringing the organism out of diapause. The latter feature is essential in distinguishing diapause as a different phenomenon from other forms of dormancy, such as stratification and hibernation. Activity levels of diapausing stages can vary considerably among species. Diapause may occur in a completely immobile stage, such as the pupae and eggs, or it may occur in very active stages that undergo extensive migrations, such as the adult Monarch butterfly, Danaus plexippus. In cases where the insect remains active, feeding is reduced and reproductive development is slowed or halted. CarotenoidFrom Wikipedia, the free encyclopedia
The orange ring surroundingGrand Prismatic Spring is due to carotenoid molecules, produced by mats of algae and bacteria.Carotenoids, also called tetraterpenoids, are organic pigments that are found in the chloroplasts and chromoplasts of plants and some other photosynthetic organisms, including some bacteria and some fungi. Carotenoids can be produced from fats and other basic organic metabolic building blocks by all these organisms. The only animals known to produce carotenoids are aphids and spider mites, which acquired the ability and genes from fungi.[1] Carotenoids from the diet are stored in the fatty tissues of animals, and exclusively carnivorous animals obtain the compounds from animal fat. There are over 600 known carotenoids; they are split into two classes, xanthophylls (which contain oxygen) andcarotenes (which are purely hydrocarbons, and contain no oxygen). All derivatives of tetraterpenes, meaning that they are produced from 8 isoprene molecules and contain 40 carbon atoms. In general, carotenoids absorb wavelengths ranging from 400-550 nanometers (violet to green light). This causes the compounds to be deeply colored yellow, orange, or red. Carotenoids are the dominant pigment in autumn leaf coloration of about 15-30% of tree species, but many plant colors, especially reds and purples, are due to other classes of chemicals. Carotenoids serve two key roles in plants and algae: they absorb light energy for use in photosynthesis, and they protect chlorophyll from photodamage.[2]Carotenoids that contain unsubstituted beta-ionone rings (including beta-carotene, alpha-carotene, beta-cryptoxanthin and gamma-carotene) have vitamin Aactivity (meaning that they can be converted to retinol), and these and other carotenoids can also act as antioxidants. In the eye, certain other carotenoids (lutein, astaxanthin,[3] and zeaxanthin) apparently act directly to absorb damaging blue and near-ultraviolet light, in order to protect the macula of the retina, the part of the eye with the sharpest vision. Contents [hide]
Biosynthesis[edit]CRT is the gene cluster responsible for the biosynthesis of carotenoids. Properties[edit]Main articles: carotenes and xanthophyllsCarotenoids belong to the category of tetraterpenoids (i.e., they contain 40 carbon atoms, being built from four terpene units each containing 10 carbon atoms). Structurally, carotenoids take the form of a polyene hydrocarbon chain which is sometimes terminated by rings, and may or may not have additional oxygen atoms attached.
Their color, ranging from pale yellow through bright orange to deep red, is directly linked to their structure. Xanthophylls are often yellow, hence their class name. The double carbon-carbon bonds interact with each other in a process called conjugation, which allows electrons in the molecule to move freely across these areas of the molecule. As the number of conjugated double bonds increases, electrons associated with conjugated systems have more room to move, and require less energy to change states. This causes the range of energies of light absorbed by the molecule to decrease. As more frequencies of light are absorbed from the short end of the visible spectrum, the compounds acquire an increasingly red appearance. Carotenoids are usually lipophilic due to the presence of long unsaturated aliphatic chains as in some fatty acids. The physiological absorption of these fat-soluble vitamins in humans and other organisms depends directly on the presence of fats and bile salts.[5] Physiological effects[edit]Supplements[edit]A 2014 review found that antioxidant supplements (including carotenoids) do not confer any health benefit and appear to increase the risk of getting certain cancers.[6] In foods[edit]Reviews of epidemiological studies seeking correlations between carotenoid consumption in food and clinical outcomes have come to various conclusions:
Simplified carotenoid synthesispathway.Plant colors[edit]The most common carotenoids include lycopene and the vitamin A precursor β-carotene. In plants, the xanthophylllutein is the most abundant carotenoid and its role in preventing age-related eye disease is currently under investigation. Lutein and the other carotenoid pigments found in mature leaves are often not obvious because of the masking presence of chlorophyll. When chlorophyll is not present, as in autumn foliage, the yellows and oranges of the carotenoids are predominant. For the same reason, carotenoid colors often predominate in ripe fruit after being unmasked by the disappearance of chlorophyll. Carotenoids give the characteristic color to carrots, corn, canaries, and daffodils, as well as egg yolks, rutabagas,buttercups, and bananas. Carotenoids are responsible for the brilliant yellows and oranges that tint deciduous foliage (such as dying autumn leaves) of certain hardwood species as hickories, ash, maple, yellow poplar, aspen, birch, black cherry, sycamore,cottonwood, sassafras, and alder. Carotenoids are the dominant pigment in autumn leaf coloration of about 15-30% of tree species.[14] However, the reds, the purples, and their blended combinations that decorate autumn foliage usually come from another group of pigments in the cells called anthocyanins. Unlike the carotenoids, these pigments are not present in the leaf throughout the growing season, but are actively produced towards the end of summer.[15] Aroma chemicals[edit]Products of carotenoid degradation such as ionones, damascones and damascenones are also important fragrance chemicals that are used extensively in theperfumes and fragrance industry. Both β-damascenone and β-ionone although low in concentration in rose distillates are the key odor-contributing compounds in flowers. In fact, the sweet floral smells present in black tea, aged tobacco, grape, and many fruits are due to the aromatic compounds resulting from carotenoid breakdown. Disease[edit]Some carotenoids are produced by bacteria to protect themselves from oxidative immune attack. The golden pigment that gives some strains ofStaphylococcus aureus their name (aureusis = golden) is a carotenoid called staphyloxanthin. This carotenoid is a virulence factor with an antioxidant action that helps the microbe evade death by reactive oxygen species used by the host immune system.[16] Question of synthesis in the corpus luteum[edit]Following a 1968 report that beta-carotene was synthesized in laboratory conditions in slices of corpus luteum from cows, an organ known to concentrate beta-carotene (hence its color and name), attempts have been made to replicate these findings, but have not succeeded. The idea is not presently accepted by the scientific community.[17] Rather, the mammalian corpus luteum, like the macula lutea in the retina of the mammalian eye, merely concentrates carotenoids from the diet. Artificial synthesis[edit]Microorganisms can be genetically modified[18] to produce certain C40 carotenoids, including lycopene and beta carotene.[19] Naturally occurring carotenoids[edit]
Choice experiment 1. Male Tetranychus urticae behaviour when presented with one female (♀) cadaver in stage 1 and one healthy ♀ quiescent deutonymph on leaf discs in a Petri dish. Stage 1=Dry non-sporulating cadaver killed by Neozygites floridana. (A) Leaf disc choice index (mean+SE) tested at three male (♂) densities. Index>0.5 indicates a preference for the leaf disc with cadaver. (B) Proportion of Petri dishes in which touching and guarding the two females were observed at least once tested at three ♂ densities.
Fatal attraction: Male spider mites prefer females killed by the mite-pathogenic fungus Neozygites floridana - Scientific Figure on ResearchGate. Available from: https://www.researchgate.net/275058361_fig2_Choice-experiment-1-Male-Tetranychus-urticae-behaviour-when-presented-with-one-female [accessed 5 Apr, 2016]
Plants respond to herbivory through various morphological, biochemicals, and molecular mechanisms to counter/offset the effects of herbivore attack. The biochemical mechanisms of defense against the herbivores are wide-ranging, highly dynamic, and are mediated both by direct and indirect defenses. The defensive compounds are either produced constitutively or in response to plant damage, and affect feeding, growth, and survival of herbivores. In addition, plants also release volatile organic compounds that attract the natural enemies of the herbivores. These strategies either act independently or in conjunction with each other. However, our understanding of these defensive mechanisms is still limited. Induced resistance could be exploited as an important tool for the pest management to minimize the amounts of insecticides used for pest control. Host plant resistance to insects, particularly, induced resistance, can also be manipulated with the use of chemical elicitors of secondary metabolites, which confer resistance to insects. By understanding the mechanisms of induced resistance, we can predict the herbivores that are likely to be affected by induced responses. The elicitors of induced responses can be sprayed on crop plants to build up the natural defense system against damage caused by herbivores. The induced responses can also be engineered genetically, so that the defensive compounds are constitutively produced in plants against are challenged by the herbivory. Induced resistance can be exploited for developing crop cultivars, which readily produce the inducible response upon mild infestation, and can act as one of components of integrated pest management for sustainable crop production. Keywords: Plant defense, herbivory, direct defense, indirect defense, biotic stress, abiotic stress Plants and insects have been living together for more than 350 million years. In co- evolution, both have evolved strategies to avoid each other’s defense systems. This evolutionary arms race between plants and insects has resulted in the development of an elegant defense system in plants that has the ability to recognize the nonself molecules or signals from damaged cells, much like the animals, and activates the plant immune response against the herbivores.1-3 To counter the herbivore attack, plants produce specialized morphological structures or secondary metabolites and proteins that have toxic, repellent, and/or antinutitional effects on the herbivores.4-6 Plants confront the herbivores both directly by affecting host plant preference or survival and reproductive success (direct defense), and indirectly through other species such as natural enemies of the insect pests (indirect defense).1,7,8 Direct defenses are mediated by plant characteristics that affect the herbivore’s biology such as mechanical protection on the surface of the plants (e.g., hairs, trichomes, thorns, spines, and thicker leaves) or production of toxic chemicals such as terpenoids, alkaloids, anthocyanins, phenols, and quinones) that either kill or retard the development of the herbivores.9 Indirect defenses against insects are mediated by the release of a blend of volatiles that specifically attract natural enemies of the herbivores and/or by providing food (e.g., extra floral nectar) and housing to enhance the effectiveness of the natural enemies.8 Research on plant-herbivore interactions is one of the most important and multidisciplinary undertakings in plant biology involving various disciplines to describe chemical and ecological processes influencing the outcome of plant - herbivore interactions. Our understanding of how plants communicate with their neighbors, symbionts, pathogens, herbivores, and with their personal “bodyguards”- the natural enemies, both above and below ground, via chemical signals, is still in its infancy. This is an enthralling area from an ecological point of view, and has a great potential for utilization in crop protection. Understanding the nature of gene expression of the plant defensive traits will have a tremendous application in designing crop plants with better protection against the herbivores. This in turn will reduce the need for use of harmful pesticides for insect control. However, the arms race between plants and herbivores will continue, and herbivores could co-evolve in response to the resistant plant genotypes. Knowledge of the complex chemical plant-herbivore interactions is required to optimize the production of new crops. Host plant defenses against insectsPlants respond to herbivore attack through an intricate and dynamic defense system that includes structural barriers, toxic chemicals, and attraction of natural enemies of the target pests (Fig. 1).1,9,10 Both defense mechanisms (direct and indirect) may be present constitutively or induced after damage by the herbivores. Induced response in plants is one of the important components of pest control in agriculture, and has been exploited for regulation of insect herbivore population.1,11,12 Over the past few decades, considerable progress has been made in studying induced responses in plants against different stresses, and has become an important topic in evolutionary biology and ecology. Although induced responses have some metabolic costs,13 they are very important when aimed at alleviating the stress of immediate concern, as most of these chemicals are produced in response to herbivore attack.14,15 Induced defenses make the plants phenotypically plastic, and thereby, decrease the chances of the attacking insects to adapt to the induced chemicals.1,12 Changes in defensive constituents of a plant on account of insect attack develop unpredictability in the plant environment for insect herbivores, which in turn, affects the fitness and behavior of the herbivores.5,6,14 If induced response occurs very early, it is of great benefit to the plant, and reduces the subsequent herbivore and pathogen attack, besides improving overall fitness of the plant.12 Plants with high variability in defensive chemicals exhibit a better defense compared with those with moderate variability.5,6 Progress in insect-plant interactions has improved our understanding of the evolution of defensive approaches exploited deployed by the plants against herbivory;10 however, the underlying mechanisms of defense are less clearly understood Direct defensesPlant structural traits such as leaf surface wax, thorns or trichomes, and cell wall thickness/ and lignification form the first physical barrier to feeding by the herbivores, and the secondary metabolites such act as toxins and also affect growth, development, and digestibility reducers form the next barriers that defend the plant from subsequent attack.9,16 Moreover, synergistic effect among different defensive components enhances the defensive system of plants against the herbivores invaders. In tomato, alkaloids, phenolics, proteinase inhibitors (PIs), and the oxidative enzymes when ingested separately result in a reduced affect, but act together in a synergistic manner, affecting the insect during ingestion, digestion and metabolism.17 InNicotiana attenuata (Torr. ex Watson), trypsin proteinase inhibitors and nicotine expression, contributed synergistically to the defensive response against Spodoptera exigua (Hub.).15 The role of morphological and biochemical constituents in host plant resistance (HPR), and induced responses to insect damage will be discussed below. Morphological structuresPlant structures are the first line of defense against herbivory, and play an important role in host plant resistance (HPR) to insects. The first line of plant defense against insect pests is the erection of a physical barrier either through the formation of a waxy cuticle,9,16 and/or the development of spines, setae, and trichomes.18,19 Structural defenses includes morphological and anatomical traits that confer a fitness advantage to the plant by directly deterring the herbivores from feeding,16 and range from prominent protrubances on a plant to microscopic changes in cell wall thickness as a result of lignification and suberization.9,19 Structural traits such as spines and thorns (spinescence), trichomes (pubescence), toughened or hardened leaves (sclerophylly), incorporation of granular minerals into plant tissues, and divaricated branching (shoots with wiry stems produced at wide axillary angles) play a leading role in plant protection against herbivory.9,19,20 Sclerophylly refers to the hardened leaves, and plays an active role in plant defense against herbivores by reducing the palatability and digestibility of the tissues, thereby, reducing the herbivore damage.9,21 Spinescence includes plant structures such as spines, thorns and prickles. It has been reported to defend the plants against many insects.9 Pubescence consists of the layer of hairs (trichomes) extending from the epidermis of the above ground plant parts including stem, leaves, and even fruits, and occur in several forms such as straight, spiral, stellate, hooked, and glandular.9 Chamarthi et al.20 reported that leaf glossiness, plumule and leaf sheath pigmentation were responsible for shoot fly Atherigona soccata (Rondani) resistance in sorghum Sorghum bicolor (L.) (Moench). TrichomesTrichomes play an imperative role in plant defense against many insect pests and involve both toxic and deterrent effects.20,21 Trichome density negatively affects the ovipositional behavior, feeding and larval nutrition of insect pests.21 In addition, dense trichomes affect the herbivory mechanically, and interfere with the movement of insects and other arthropods on the plant surface, thereby, reducing their access to leaf epidermis.16 These can be, straight, spiral, hooked, branched, or un-branched and can be glandular or nonglandular.9 Glandular trichomes secrete secondary metabolites including flavonoids, terpenoids, and alkaloids that can be poisonous, repellent, or trap insects and other organisms, thus forming a combination of structural and chemical defense.9,18 Induction of trichomes in response to insect damage has been reported in many plants.22 This increase in trichome density in response to damage can only be observed in leaves developing during or subsequent to insect attack, since the density of trichomes of existing leaves does not change.16 Dalin and Bjorkman23reported that damage by adult leaf beetles, Phratora vulgatissima L. in Salix cinerea L. induced higher trichome density in the new leaves developing thereafter. Likewise, increase in trichome density in S. cinerain response to coleopteran damage has also been reported.24 Increase in trichome density after insect damage has also been reported in Lepidium virginicum L. and Raphanus raphanistrum L.22 In black mustard, trichomes density and glucosinolate levels were elevated after feeding by Pieris rapae (L.).25 Trichome density increased in Alnus incana Moench as a result of damage by beetles.26 The increase in trichome density in response to herbivory is typically between 25 to 100%, however, there are cases where 500 – 1000% increase in trichome density has also been reported. Changes in trichome density occur within days or weeks after insect damage.22-24 Furthermore, change in relative proportion of glandular and non-glandular trichomes is also induced by herbivory.22 A positive correlation has been observed between natural enemies’ abundance and trichome density. Trichome exudates also serve as extra floral nectar (EFN) for scelonid egg parasitoid, of squash bugs, Gryon pennsylvanicum.27 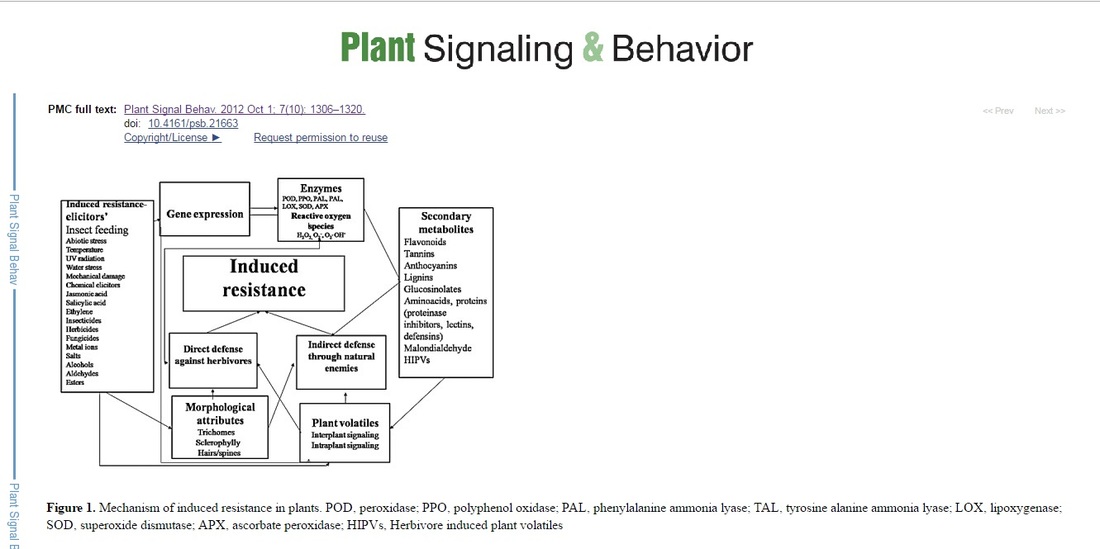 Secondary metabolites and plant defenseSecondary metabolites are the compounds that do not affect the normal growth and development of a plant, but reduce the palatability of the plant tissues in which they are produced.1 The defensive (secondary) metabolites can be either constitutive stored as inactive forms or induced in response to the insect or microbe attack. The former are known as phytoanticipins and the latter as phytoalexins. The phytoanticipins are mainly activated by β-glucosidase during herbivory, which in turn mediate the release of various biocidal aglycone metabolites.28 The classic examples of phytoanticipins are glucosinolates that are hydrolyzed by myrosinases (endogenous β-thioglucoside glucohydrolases) during tissue disruption. Other phytoanticipins include Benzoxazinoids (BXs), which are widely distributed among Poaceae. Hydrolyzation of BX-glucosides by plastid-targeted β-glucosidases during tissue damage leads to the production of biocidal aglycone BXs, which play an important role in plant defense against insects.28 Phytoalexins include isoflavonoids, terpenoids, alkaloids, etc., that influence the performance and survival of the herbivores.29 The secondary metabolites not only defend the plants from different stresses, but also increase the fitness of the plants. It has been reported that maize HPR to corn earworm, Helicoverpa zea (Boddie) is mainly due to the presence of the secondary metabolites C-glycosyl flavone maysin [2”- O - a –L-rhamnosyl- 6- C - (6-deoxy-xylo -hexos-4-ulosyl) luteolin] and the phenylpropanoid product, chlorogenic acid.30 Compound, 4, 4- dimethyl cyclooctene has been found to be responsible for shoot fly A. soccata resistance in sorghum S. bicolor.31 Secondary metabolites have been primarily studied as the mediators of direct defense, however much is to be done to reveal the unidentified or emerging signaling pathways. Mass spectrometry used for the secondary metabolite profiling and gene expression analysis by high-throughput sequencing has made this field more exciting and cost-effective. Study on secondary metabolites could lead to the identification of new signaling molecules involved in plant resistance against herbivores and other stresses. Ultimately genes and enzymes involved in the biosynthesis of these metabolites could be identified. Role of some of the secondary metabolites in plant defense will be discussed below. Plant phenolicsAmong the secondary metabolites, plant phenols constitute one of the most common and widespread group of defensive compounds, which play a major role in HPR against herbivores, including insects.4-6,18 Phenols act as a defensive mechanism not only against herbivores, but also against microorganisms and competing plants. Qualitative and quantitative alterations in phenols and elevation in activities of oxidative enzyme in response to insect attack is a general phenomenon.5,6,32 Lignin, a phenolic heteropolymer plays a central role in plant defense against insects and pathogens.32 It limits the entry of pathogens by blocking physically or increasing the leaf toughness that reduces the feeding by herbivores, and also decreases the nutritional content of the leaf.33 Lignin synthesis has been found to be induced by herbivory or pathogen attack and its rapid deposition reduce further growth of the pathogen or herbivore fecundity.33 Increase in expression of lignin associated genes (CAD/CAD-like genes) in plants infected with pests and pathogens have been documented.32 Oxidation of phenols catalyzed by polyphenol oxidase (PPO) and peroxidase (POD) is a potential defense mechanism in plants against herbivorous insects. Quinones formed by oxidation of phenols bind covalently to leaf proteins, and inhibit the protein digestion in herbivores.34 In addition, quinones also exhibit direct toxicity to insects.17,34 Alkylation of amino acids reduces the nutritional value of plant proteins for insects, which in turn negatively affects the insect growth and development.34 Phenols also play an important role in cyclic reduction of reactive oxygen species (ROS) such as superoxide anion and hydroxide radicals, H2O2, and singlet oxygen, which in turn activate a cascade of reactions leading to the activation of defensive enzymes.35 Simple phenolics (salicylates) act as antifeedant to insect herbivores such as Operophtera brumata (L.) in Salix leaves, and there is a negative correlation between the salicylate levels and the larval growth, however, salicylic acid (SA) is much more important as phytohormone than as deterrent.36 FlavonoidsFlavonoids play a central role in various facets of plant life especially in plant-environment interactions.37These defend plants against various biotic and abiotic stresses including UV radiations, pathogens and insect pests.37 Flavonoids are cytotoxic and interact with different enzymes through complexation. Both flavonoids and isoflavonoids protect the plant against insect pests by influencing the behavior, and growth and development of insects.36 In addition, flavonoids scavenge the free radicals including ROS, and reduce their formation by chelating the metals.37 Flavonoids are divided into various classes that include anthocyanins, flavones, flavonols, flavanones, dihydroflavonols, chalcones, aurones, flavan, and proanthocyanidins.37More than 5,000 flavonids have been reported in plants. A number of flavones such as flavonols, flavones, proanthocyanidins, flavan 3-ols, flavonones, flavans, and isoflavonoids have been investigated as feeding deterrents against many insect pests. Flavonoids such as flavones 5 -hydroxyisoderricin,7- methoxy-8- (3- methylbutadienyl) –flavanone and 5-methoxyisoronchocarpin isolated from Tephrosia villosa (L.), T. purpurea (L.), and T. vogelii Hook, respectively have been found as feeding deterrents against Spodoptera exempta (Walk.), and Spodoptera littoralis Bios.38 Overexpressing a transcription factor controlling flavonoid production in Arabidopsis has been reported to confer resistance against Spodoptera frugiperda(J.E. Smith).39 Angustone A, licoisoflavone B, angustone B, and angustone C. Isoflavones, licoisoflavone A, luteone, licoisoflavone B, and wighteone have been found to be not only feeding deterrents to insects, but also have antifungal activity against the fungi, Colletotrichum gloeosporiode (Penz.) and Cladosporium cladosporioides (Fres.).40 Isoflavonoids (judaicin, judaicin-7-O-glucoside, 2-methoxyjudaicin, and maackiain) isolated from the wild relatives of chickpea act as antifeedant against Helicoverpa armigera(Hubner) at 100 ppm. Judaicin and maackiain were also found to be deterrent to S. littoralis and S. frugiperda, respectively.41 Cyanopropenyl glycoside and alliarinoside strongly inhibit feeding by the native American butterfly, Pieris napi oleracea L., while a flavone glycoside, isovitexin-6”-D-β-glucopyranoside acts as a direct feeding deterrent to the late instars.42 TanninsTannins have a strong deleterious effect on phytophagous insects and affect the insect growth and development by binding to the proteins, reduce nutrient absorption efficiency, and cause midgut lesions.18,43,44 Tannins are astringent (mouth puckering) bitter polyphenols and act as feeding deterrents to many insect pests. They precipitate proteins nonspecifically (including the digestive enzymes of herbivores), by hydrogen bonding or covalent bonding of protein –NH2 groups. In addition, tannins also chelate the metal ions, thereby reducing their bioavailability to herbivores. When ingested, tannins reduce the digestibility of the proteins thereby decrease the nutritive value of plants and plant parts to herbivores. Role of tannins in plant defense against various stresses and their induction in response to insect damage has been studied in many plants.44 For example, e.g., in Populus species,45 and in Pinus sylvestris L.46 However, no effect of herbivore damage on tannin content was observed in Quercus serrata (Thunb.)47 and Betula pendulaRoth.48 Like proteinase inhibitors and oxidative enzymes, tannins have been reported to be systemically induced in neighboring leaves of the damaged plant.45 Condensed tannins are oligomeric or polymeric flavonoids, also known as proanthocyanidins. They have diverse structures and functions. They act as feeding deterrents against some insects such as, Lymantria dispar (L.), Euproctis chrysorrhoea (L.) and O. brumata.49,50 Condensed tannins such as (+) -catechin, (+) - gallocatechin, and vanillin in leaves of Quercus robur L. inhibited winter moth larvae, O. brumata.49Procyanindin polymers have been found as feeding deterrent to Aphis Craccivora (Koch) in groundnut.51Condensed tannins from Alaska paper birch (coated on birch leaves at 3% dry wt.) reduced the pupal mass and survival of Rheumaptera hastata (L.) larvae.52 It has been reported that induction of tannins in Populus tremuloides Michx. leaves in response to wound- and herbivore occur by transcriptional activation of the flavonoid pathway.45 Genes responsible for the production of tannins in response to wounding have been identified and are activated by the expression of a condensed tannins regulatory gene, PtMYB134, which is itself induced by damage.53 Furthermore, induction of tannin is also stimulated by light stress,14,53 and exposure to UV light in hybrid poplar.53 However, some polyphagous insect species have the ability to tolerate gallotannins, e.g., Shistocerca gregaria (Forsk.) tolerates tannins by hydrolyzing them rapidly to avoid any damaging effects by restricting the passage of tannins by adsorbing them on the thick peritrophic membrane, and by inhibiting the tannin protein complex formation by surfactants in the midgut.54 Plant defensive proteinsEcologically, in insect-plant interaction, interrelationship between two is important for the survival of the both. Insects always look for a true and healthy host plant that can provide them proper food and could be suitable for mating, oviposition and also provides food for the offsprings. The nutritional requirements of insects are similar to other animals, and any imbalance in digestion and utilization of plant proteins by the insects’ results in drastic effects on insect physiology. Alteration of gene expression under stress including insect attack leads to qualitative and quantitative changes in proteins, which in turn play an important role in signal transduction, and oxidative defense (Table 1).4,55 Many plant proteins ingested by insects are stable, and remain intact in the midgut, and also move across the gut wall into the hemolymph. An alteration in the protein’s amino acid content or sequence influences the function of that protein. Likewise, anti-insect activity of a proteolysis-susceptible toxic protein can be improved by administration of protease inhibitors (PIs), which prevent degradation of the toxic proteins, and allows them to exert their defensive function. Better understanding of protein structure and post-translational modifications contributing to stability in the herbivore gut would assist in predicting toxicity and mechanism of plant resistance proteins (PRPs). Recent advances in microarray and proteomic approaches have revealed that a wide spectrum of PRPs is involved in plant defense against herbivores.56,57 Due to diverse feeding habits of arthropods, multiple signaling pathways including jasmonic acid (JA), SA and/or ethylene (ET) regulate arthropod-inducible proteins.8 Table 1. Plant defensive proteins against insect pests
The mode of action of fenpyroximate in Tetranychus urticae Koch (two-spotted spider mite) was studied biochemically and morphologically. Biochemical assays showed that an adult female T. urticae contained 88.96 ± 10.32 pmol ATP, which rapidly decreased to 74.8, 31.2, and 2.9%, 60 min after spraying with 0.05, 0.5, and 5 μg/ml fenpyroximate, respectively. The mortalities at that time were 2, 18, and 46%, respectively. In vitro experiments using rat liver mitochondria showed that 10 μMfenpyroximate inhibited electron transport when NADH or NADH-linked substrates were used as the electron donors but not when succinate was used as the substrate. Therefore, the site of inhibition on the electron transport chain was considered to be the NADH-coenzyme Q (Co Q) reductase. Mitochondrial NADH-Co Q reductase of T. urticae was also inhibited by fenpyroximate, and the I50 value was estimated to be 0.08 μM. Transmission electron microscopy observations of T. urticae sprayed with 0.5 μg/ml fenpyroximate showed that the compound caused morphological changes in mitochondria in peripheral nerve cells such as swelling, irregular cristae arrangement, and lower matrix electron density. Similar morphological changes in mitochondria were also obvious in the ovaries and epidermal cells, but not in muscular cells or central nervous mass cells. These results indicate that the inhibition of mitochondrial NADH-Co Q reductase by fenpyroximate seems to induce a decrease in ATP contents and morphological changes in mitochondria. Ultimately, this would contribute to the acaricidal and knockdown activities against T. urticae by this compound.
Bibliography |
|
||||||





 RSS Feed
RSS Feed